Groundwater hydrochemistry—revealing the secrets of a hidden water resource
09 March 2017
Water is commonly thought of as just H2O, but groundwater contains a wide range of dissolved chemical elements from the surrounding environment. Most of these occur naturally, and many are present only in small quantities. These elements can tell us a lot about the source of the water, its quality and possible uses.
Image: Groundwater chemistry sampling on an artesian bore in South Australia. Credit: Geoscience Australia.
What is hydrochemistry?
The term hydrochemistry or ‘water chemistry’ refers to the quantities of the various chemical elements present in a particular water sample.
In most groundwater, typically only seven ions make up nearly 95 per cent of all ions present. These are calcium, magnesium, sodium, potassium, chloride, sulphate, and bicarbonate. In addition, other chemical elements known as minor and trace elements usually exist in small quantities, as do particular isotopes.
What is groundwater hydrochemistry used for?
The hydrochemistry of any groundwater sample has a distinct chemical signature. This signature reflects the sum of all processes that affected the water from the time it began as rainfall, infiltrated the soil above the water table, passed into the aquifer (body of permeable rock which contains and transmits groundwater), and travelled (sometimes over great distances and depth), to the point of sample collection or discharge from the aquifer.
Processes that affect hydrochemistry can provide a powerful tool for tracking groundwater flow and managing groundwater in a sustainable way.
Hydrochemistry can help to answer many questions, such as:
- Can this water be used for drinking, irrigation or stock and domestic needs?
- Where does this groundwater come from?
- How long ago was the groundwater replenished?
- How old is the groundwater?
Case study: the Great Artesian Basin
Hydrochemistry analysis has played a major role in increasing our understanding of groundwater processes in Australia’s largest groundwater basin—the Great Artesian Basin. It has been used to determine the rate of groundwater flow, recharge mechanisms and rates, the age of groundwater, inter-aquifer connectivity, and to delineate flow systems within the basin. This information has been integral for the development of appropriate management of important groundwater resources.
In many cases hydrochemistry analysis is the only readily accessible technique for understanding groundwater processes in remote parts of Australia.
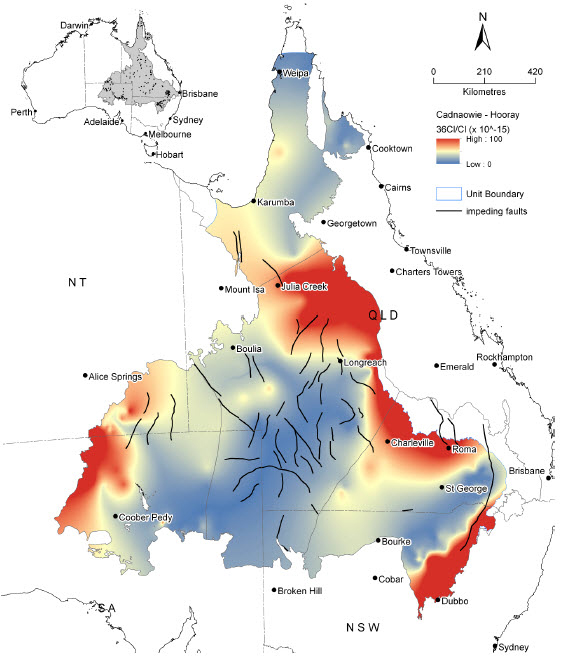
Map: Ratio of chlorine-36 to chloride in the Cadna-owie - Hooray Aquifer and equivalents in the Great Artesian Basin. High values, shown in red, can be used to identify recharge areas. Credit: CSIRO and Geoscience Australia, 2013.
Hydrochemistry data in the Australian Groundwater Explorer
The Bureau and Geoscience Australia (GA) collaborated to include hydrochemistry data for more than 500 bores across Australia in the Australian Groundwater Explorer (the Explorer).
GA generally collects a range of hydrochemistry data on a project basis including:
- common measures, such as pH and electrical conductivity;
- major and minor ions, such as sodium and calcium;
- environmental tracers, such as oxygen-18, deuterium, carbon-13;
- radioactive isotopes, such as chloride-36 and carbon-14; and
- nutrients, such as nitrate.
These data are now available in the Explorer as a table, which can be downloaded for further analysis.
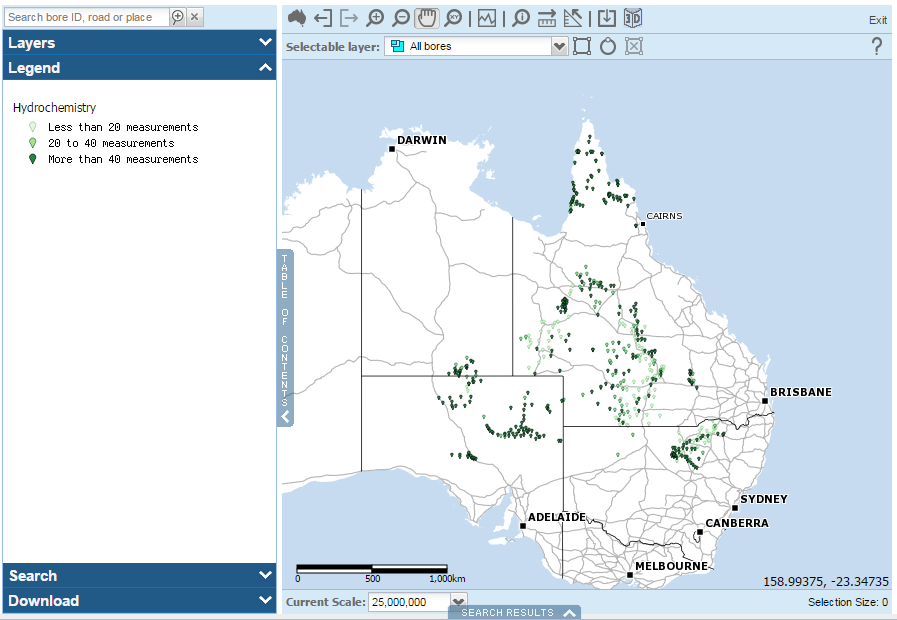
Map: GA bores with hydrochemistry data in the Explorer
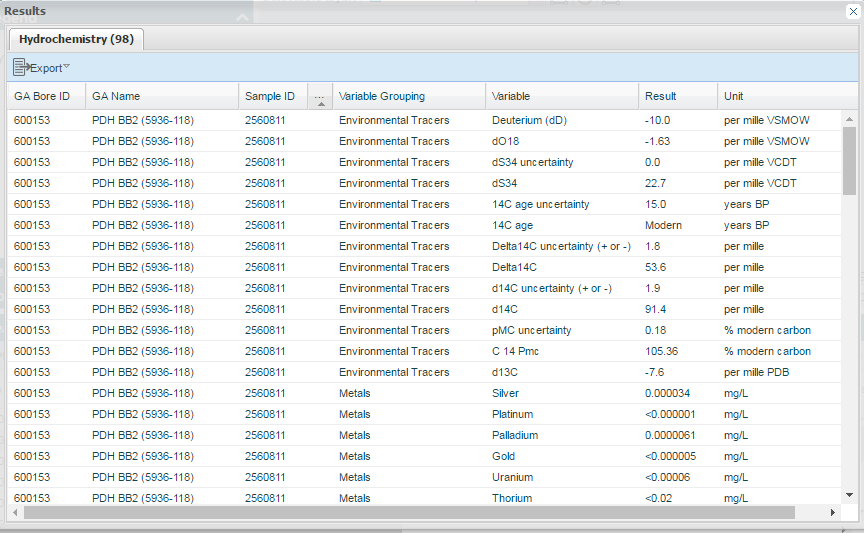
Table: Hydrochemistry data for a bore in South Australia
More information
Our groundwater products can be found at Groundwater information.

Subscribe to this blog to receive an email alert when new articles are published.


Comment. Tell us what you think of this article.
Share. Tell others.